Fluorescent LAMP Kit Biofargo
$ 150,00 Original price was: $ 150,00.$ 60,00Current price is: $ 60,00.
Product Description
-
Optimized LAMP mix, contains a fluorescent dye.
-
Real-time monitoring of the LAMP reactions using real-time PCR machine.
-
Useful in optimizing LAMP primers or reaction parameters.
-
4× concentration LAMP mix allows larger in-put sample volumes than 2× concentration mix, which can increase the sensitivity of the test.
-
High amplification efficiency, generally an order of magnitude more sensitive than PCR.
-
Isothermal amplification around 65°C, no need for expensive thermal cycling instruments.
-
Template-primer mix supplied as LAMP positive control, eliminates false negative results.
-
CAT#211222-50 is sufficient for 50 reactions of 20μL volume, and CAT#211222-500 for 500 reactions of 20μL volume.
Input Sample Requirements
-
Perform DNA purification with a positive and a negative control. For a sample number of N, perform N+2 purifications. For the two additional purifications,one is for positive control and another negative control.
Kit Contents
|
Component
|
CAT#:211222-50
|
CAT#:211222-500
|
|
4×Fluorescent LAMP Mix
|
200μL
|
1mL
|
|
Bst DNA Polymerase 2.0
|
50μL
|
250μL
|
|
Template-primer Mix
|
50μL
|
250μL
|
Storage and Handling
Required Materials not Supplied
|
No
|
Item
|
|
1
|
Prepared DNA Samples
|
|
2
|
LAMP primers targeting DNA samples
|
|
3
|
Reaction vessels compatible with instrument for LAMP
|
|
4
|
Molecular biology grade ddH2O
|
|
5
|
Real-time PCR machine
|
Procedural Guidelines
-
Perform all steps at room temperature (20–25°C) unless otherwise noted.
-
Regularly de-contaminate working areas and equipment using chlorine bleach to avoid potential carryover contamination.
-
Avoid opening reaction vessels after a LAMP reaction is completed..
-
Screening at least 10 sets of LAMP primers for optimal sensitivity and specificity before choosing a final set.
-
Use Loop primers if possible.
-
Primers should be HPLC purified.
Procedures
-
Make 20×Primer Mix. Use appropriate amount of ddH2O to dissolve the HPLC-purified LAMP primers at 100μM each. Use the 100μM stocks to make 100μL 20×Primer Mix according to the following table. If volumes other than 100μL (50μL, 200μL etc.) is desired, adjust volumes accordingly.
|
Components
to add
|
Volume
to add
|
Final Concentration
in 20×Primer Mix
|
|
100μM FIP
|
32μL
|
32 μM
|
|
100μM BIP
|
32μL
|
32 μM
|
|
100μM Loop F
|
4μL
|
8 μM
|
|
100μM Loop B
|
4μL
|
8 μM
|
|
100μM F3
|
2μL
|
4 μM
|
|
100μM B3
|
2μL
|
4 μM
|
|
ddH2O
|
24μL
|
–
|
-
Thaw all components to be used at room temperature. Vortex briefly 4×Fluorescent LAMP Mix and Template-primer Mix. Centrifuge to collect material and place on ice. Don’t vortex Bst DNA polymerase 2.0.
-
When using the kit for the first time,add all Bst DNA polymerase 2.0 to 4×Fluorescent LAMP Mix and gently invert the tube for one minute to mix thoroughly.
-
For a purification number of N+2, set up N+4 LAMP reactions as described below. Volumes listed are for 20μL reaction. For the two additional reactions, one is for LAMP Positive Control (PC) and another for LAMP Negative Control (NC) .
|
|
PC
|
NC
|
N+2
|
|
4×Fluorescent LAMP Mix
|
5μL
|
5μL
|
5μL each
|
|
20×Primer Mix
|
1μL
|
1μL
|
1μL each
|
|
Template-primer Mix
|
5μL
|
|
|
|
N+2 samples
|
|
|
1-14μL each
|
|
ddH2O
|
9μL
|
14μL
|
Add to 20μL
|
-
Seal reaction vessels.
-
Incubate at 65°C for 60 minutes in a real-time PCR machine. Collect signal in the SYBR channel in every 60 seconds.
-
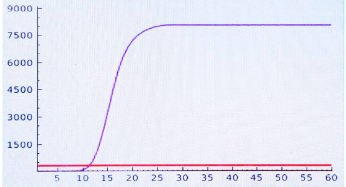
The two positive reactions should have S-shaped fluorescent signal. The two negative controls should have no S-shaped fluorescent signal.
-
If there is amplification in NC or there is no amplification in PC,the whole experiment is invalid. There is no need to analysis the results of the N+2 samples. Refer to Troubleshooting section.
-
If there is no amplification in NC or there is amplification in PC,the whole experiment is valid. Analyze and record he results of the N+2 samples. The results from fluorescent detection can be downloaded from the real-time PCR machine.
Troubleshooting
-
There is amplification in NC.
-
This result suggests possible carryover contamination in working environment and/or reagents. To avoid carryover contamination, clean working areas and equipment using a 10% chlorine bleach solution, use new (un-contaminated) reagents, primer stocks, water, etc., use UNG-LAMP Kit, perform experiments in a new (un-contaminated) room.
-
This result also suggests nonspecific amplification. To avoid nonspecific amplification, do amplification at temperature of 65°C -70°C,or put the reaction tubes to 65°C as soon as the reaction is set,or use a new set of primer for the targeted DNA region.
-
-
There is no amplification in PC
-
This result suggests failures in machine or reagents,use new machine or new set of reagents.
-
Limited Product Warranty
-
Biofargo ,Inc. warrants its products as set forth in the Biofargo ’s General Terms and Conditions of Sale at . If you have any questions, please contact Biofargo @ .
-
The information in this guide is subject to change without notice.
-
DISCLAIMER: TO THE EXTENT ALLOWED BY LAW, Biofargo INC. WILL NOT BE LIABLE FOR SPECIAL, INCIDENTAL, INDIRECT, PUNITIVE, MULTIPLE, OR CONSEQUENTIAL DAMAGES IN CONNECTION WITH OR ARISING FROM THIS DOCUMENT, INCLUDING YOUR USE OF IT.
Quick Shipping and Professional Packaging
We can offer a variety of shipping options due to our long-term relationship with UPS FedEx DHL. Our warehouse staff will package each item according to our strict specifications. Your products will be subjected to a thorough examination and will be safely packaged prior to being shipped. Everyday we deliver thousands of packages to clients across many countries. This is a sign of our determination to become the biggest online retailer in the world. There are distribution centers and warehouses in Europe as well as the USA.
Orders that contain more than 1 item are given processing time for each item.
Before shipment, all ordered items will be thoroughly inspected. Most orders are shipped within 48-hours. Delivery is expected to take between 3 and 7 days.
Returns
The stock market is always changing. It is not managed entirely by us, as we're involved with several parties such as the factory and our storage. The levels of stock can change at any given time. Be aware that it is possible for your order to run out of stock even after you have placed the order.
The policy is 30 days. However, if 30 days have passed since you purchased your item, we will not be able to offer you a return or exchange.
To be eligible for a refund the item must be in good condition and in the same state as when you received it. It should also be in the original package.
Related products
Uncategorized
Uncategorized
2mL Amber Glass Screw Thread Vials, HPLC Autosampler Vials,With Scale, 100/PK Biofargo
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
12 Well Cell Culture Plate, Flat, TC, sterile, Bulk, 10/pk, 50/cs, (712002) NSTF90073 Biofargo
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized